
Despite the widespread use of lithium-ion batteries, there is still an urgent need for accurate modeling techniques to understand their complex behavior and optimize their performance. By providing a simplified but efficient representation of lithium-ion batteries, equivalent circuit modeling has become a valuable way to meet this requirement. Despite their small size, lithium-ion batteries are complex electrochemical systems that involve complex processes such as ion diffusion, embedding, and side reactions.
Equivalent circuit modeling provides a practical solution by representing the battery as an electrical network composed of passive components. These elements, including resistors, capacitors, and ideal voltage sources, capture the fundamental characteristics of the battery's behavior. By accurately simulating the behavior of batteries under different operating conditions, equivalent circuit modeling enables engineers and researchers to optimize battery design, improve performance, and extend battery life.
When utilized effectively, equivalent circuit modeling gives engineers insight into the internal behavior of lithium-ion batteries. This understanding allows them to optimize various aspects, such as battery design, material selection, and charging/discharging protocols. In addition, the equivalent circuit model (ECM), as the basis of battery management system development, can realize real-time monitoring, state estimation and control of lithium-ion batteries in practical applications.
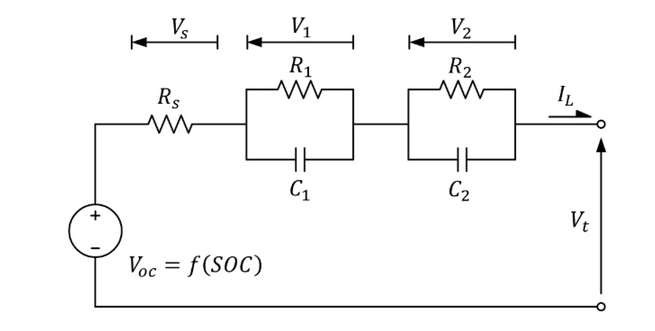
Network connection of resistance-capacitance
ECM is a useful dynamic representation of a battery that can be enhanced to capture nonlinear properties. The simplest ECM consists of a single resistance-capacitance (RC) network and cannot accurately describe the charging and discharging process. However, as shown in Figure 2, the accuracy of the model can be significantly improved by introducing multiple RC networks, such as the popular second-order RC network (2RC). This improvement can more effectively describe the nonlinear behavior of the battery. In addition, increasing the number of RC networks in the ECM provides more efficient simulations while maintaining the convenience of determining model parameters.
Figure 1: Experiment 2RC model circuit
At this stage, it was noted that increasing the number of RC devices in the circuit could improve model accuracy. However, a trade-off was observed between accuracy and the disadvantages of increased power consumption and longer computation times.
To build a battery model, it is important to determine the appropriate model parameters. By studying the relationship between state of charge (SOC) and open circuit voltage (OCV) under specific laboratory conditions, off-line identification methods were developed to determine these parameters.
Understand SOC and OCV
The battery voltage is directly affected by the SOC, with the highest voltage when the battery is full and the lowest voltage when the battery is empty. The relationship between OCV and SOC is specific to the battery technology used and is critical to accurately estimating the battery voltage and SOC. To establish this OCV-SOC relationship, two sets of experiments were conducted.
The first group involved three discharge tests that reduced the SOC from 100% to 20% each time. A constant current of 7.3A was subtracted and the voltage was measured after a 20-hour quiescent period. This stabilizes the battery voltage, providing the best approximation of the OCV. SOC levels were measured at 100%, 80%, 60% and 40%.
The second set of experiments consisted of three charging tests starting at 40% of the initial SOC. The same current strength used in the discharge test is added back to the battery for the same duration. A quiescent period is introduced before voltage measurement. These experiments produced data points used to construct the OCV-SOC curve of the battery pack.
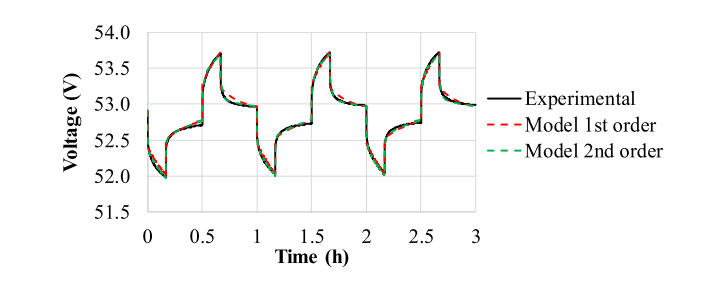
Figure 2: Battery voltage measurement and model estimation
Additional experiments were performed to determine the resistance (R) and capacitance (C) model parameters. Three charge and discharge cycles were performed, starting with a constant current discharge of 7.3A for 10 minutes. After a 20-minute quiescent period, the battery is charged at the same current for 10 minutes. An additional 20-minute quiescence period was performed before the cycle was repeated. The model parameters are adjusted by minimizing the difference between the measurements for each SOC and calculating the battery voltage.
Figure 2 shows the superior performance of the second-order model compared to the first-order model. The average relative error of the second-order model is 0.03% and the maximum relative error is 0.71%, while the average relative error of the first-order model is 0.06% and the maximum relative error is 1.04%. Nevertheless, establishing a direct correlation between the set parameters of a second-order model and the SOC proved to be a challenging task.
Different parameters are established for the charging and discharging processes to address their inherent differences. The accuracy of the model was thoroughly evaluated by using multiple discharge-charge cycles with different current values. Each cycle consists of a 10-minute discharge phase, followed by a 20-minute quiescent period, a 10-minute charging phase and another 20-minute quiescent period. This comprehensive test approach is designed to ensure high accuracy and reliability of the model under a variety of operating conditions.
Establish model parameters
In the experiments conducted, a 2RC circuit model was formulated to analyze the electrochemical model. The choice of model parameters R1, R2, C1 and C2 was carefully considered. The OCV is then calculated using the pulse test data. To accurately determine the parameters, sophisticated identification techniques such as multiple linear regression (MLR), exponential curve fitting (ECF), and Simulink Design Optimization Tool (SDOT) are employed.
Figure 3:2RC electrochemical model for MLR(solid line), ECF(dashed line), and SDOT(dashed line) parameters
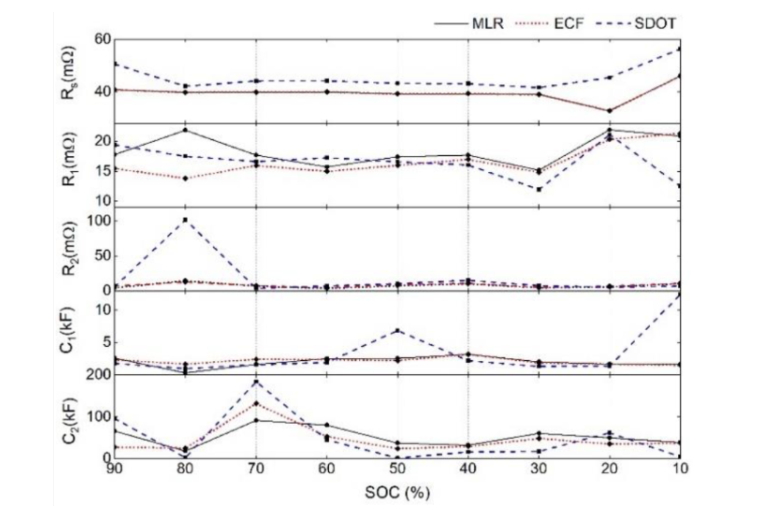
The estimated parameters for MLR, ECF, and SDOT are shown in Figure 3. MLR and ECF share the same internal resistance (Rs) value because they both calculate this value using pulse test voltage response data. However, SDOT uses different methods, resulting in different estimation parameters. At low SOC levels (0% to 30%), resistance values (Rs and R1) tend to increase. The polarization parameters exhibit an interesting tradeoff: the significant increase in polarization resistance (Ri) seen in SDOT is compensated by a decrease in polarization capacitance (Ci), and vice versa. Notably, the parameters remain relatively stable between 30% and 60% SOCs, changing significantly in the 0% to 30% and 70% to 100% SOC ranges due to the dominance of nonlinearities in the OCV-SOC relationship.
Figure 4: Pulse discharge test cell terminal voltages show the relative inaccuracies of the three estimates.
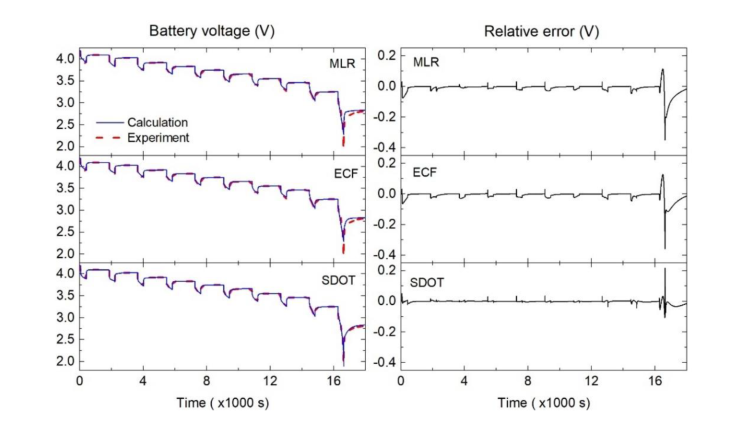
Figure 4 shows the estimated battery end voltage during the pulse test, showing excellent agreement between the prediction and experimental data, especially SDOT. Relative error indicates that the difference between simulation and experiment is close to zero, but the SOC level is low (< 10%SOC).
Conclusion
In this paper, an experiment is conducted to investigate parameter identification and modeling of lithium-ion batteries. The study explored different approaches, including MLR, ECF, and SDOT algorithms, which vary in complexity and mathematical background. The SDOT algorithm shows the best fit for pulse testing, while MLR and ECF show considerable accuracy in constant-current applications, and the amount of computation associated with SDOT may limit its application in electric vehicles.
The proposed approach involves the use of an electrochemical model that takes into account the nonlinear relationship between SOC and OCV, as well as the differences between charging and discharging processes. Due to the difficulty in correlating second-order model parameters to SOCs, the study recommends the use of first-order models. This approach shows the promise of accurately modeling lithium-ion batteries of various types and capacities. The findings contribute to a better understanding of battery behavior and provide valuable insights for energy storage and electric vehicle applications.
About US
Heisener Electronic is a famous international One Stop Purchasing Service Provider of Electronic Components. Based on the concept of Customer-orientation and Innovation, a good process control system, professional management team, advanced inventory management technology, we can provide one-stop electronic component supporting services that Heisener is the preferred partner for all the enterprises and research institutions.
